 The change could be written: H 2 O ( l) H 2 O ( g) Chemical: The dark grey nail changes color to form an orange flaky substance (the rust) this must be a chemical change. When water boils, no bonds are broken or formed. The green mixture is still green and the colorless solution is still colorless. Physical: boiling and melting are physical changes. Physical: because none of the properties changed, this is a physical change. Students check their work by color coding answers on a grid. Different smell or taste (do not taste your chemistry experiments, though!).\): (a) Copper and nitric acid undergo a chemical change to form copper nitrate and brown, gaseous nitrogen dioxide. Here are 27 easy-prep, memorable and hands-on activities for middle schoolers to defy their misconceptions about physical and chemical changes and master the basics 1. The following reaction occurs: Fe + O 2 Fe 2 O 3. Students sort through a total of fifty six statements describing either a physical change or chemical change.Bubbles are formed (but the substance is not boiling-you made a substance that is a gas at the temperature of the beginning materials, instead of a liquid).Unexpected color changes (a substance with a different color is made, rather than just mixing the original colors together).Physical changes do not alter the identity of a substance. Temperature changes (either the temperature increases or decreases). A change in physical properties is called a physical change.Some physical properties, such as density and color, may be observed without changing the physical. Familiar examples of physical properties include density, color, hardness, melting and boiling points, and electrical conductivity. This quiz and worksheet combination will help you test your understanding of how. A physical property is a characteristic of matter that is not associated with a change in its chemical composition. 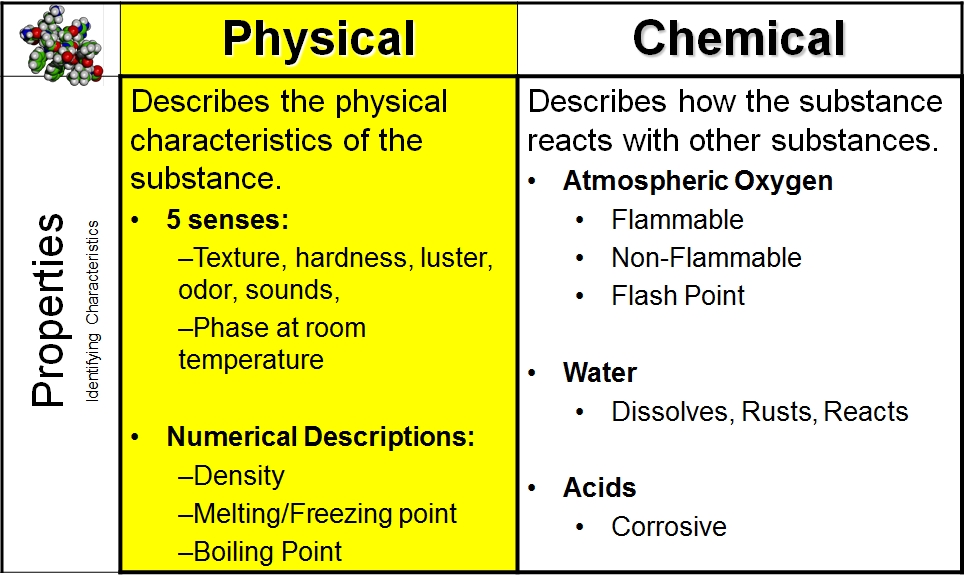
Observations that help to indicate chemical change include: The physical and chemical properties of matter can be used to describe anything that takes up space and has mass. 
Some of the evidence for chemical change will involve the energy changes that occur in chemical changes, but some evidence involves the fact that new substances with different properties are formed in a chemical change. We have to make other observations to indicate that a chemical change has happened. We can't actually see molecules breaking and forming bonds, although that's what defines chemical changes. Design an experiment to test properties of different combinations of materials. Create your own free activity from our activity creator. 
Explore the new properties exhibited when new materials are made from combinations of two or more original materials. Do you want to stay in the Top 10 of this activity Log in to identify yourself. \): Burning of wax to generate water and carbon dioxide is a chemical reaction. Make observations and cite evidence to identify changes as chemical or physical. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
